Reactivity
Towards Mechanism
Basic ideas
Reactivity is mostly governed by bond strengths as well as atoms in molecules being either electron-rich or electron-poor; electron-rich atoms (i.e. bases or nucleophiles) attack electron-poor atoms (i.e. Lewis acids or electrophiles). Atoms and molecules want to become more stable and have a variety of pathways (mechanisms) to low energy states; some pathways are reversible, which leads to equilibria and the application of Le Chatelier’s principle. Kinetics relates to how fast a reaction occurs and how high activation barriers will be; simple ideas like a crowded environment means slower reactions while accessible environments (i.e. easier to get to) means faster reactions and lower Eact.
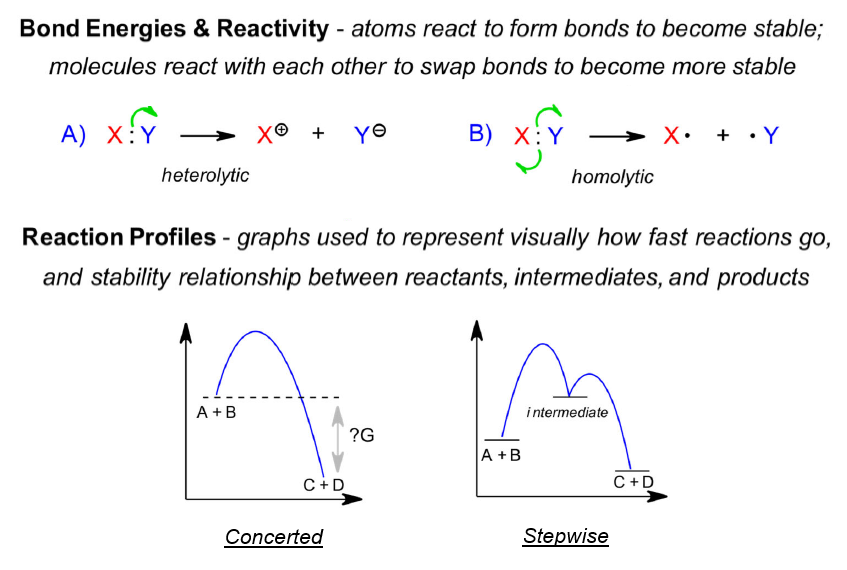
As in General Chemistry, molecules are going to react with other molecules so that the whole system becomes more stable. Weak bonds get swapped for stronger bonds while charge (positive or negative) finds its way to the best location possible. These ideas are used throughout the Organic sequence to explain how chemicals change.
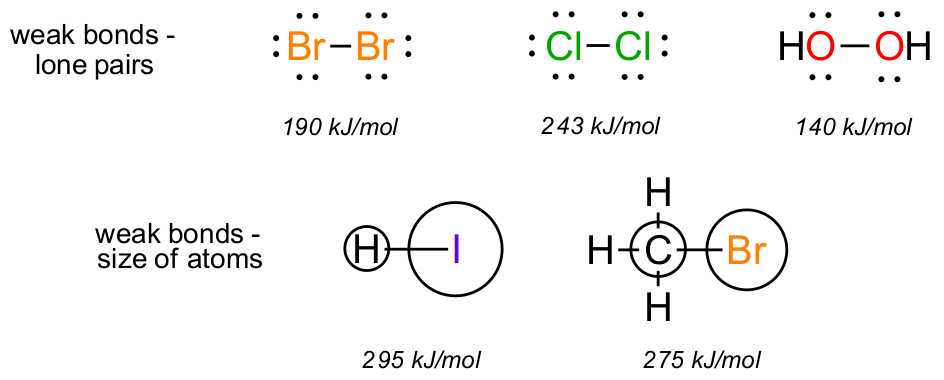
What constitutes a reactive molecule? Weak bonds are a good place to start. Numerous reactions feature reagents such as Br-Br, HO-OH and similar which have atoms with multiple lone pairs directly next to each other. These bonds will always be broken along the way during reactions to give more stable outcomes. Likewise, when atoms of quite different size bond together those bonds tend to be weak and easily broken.
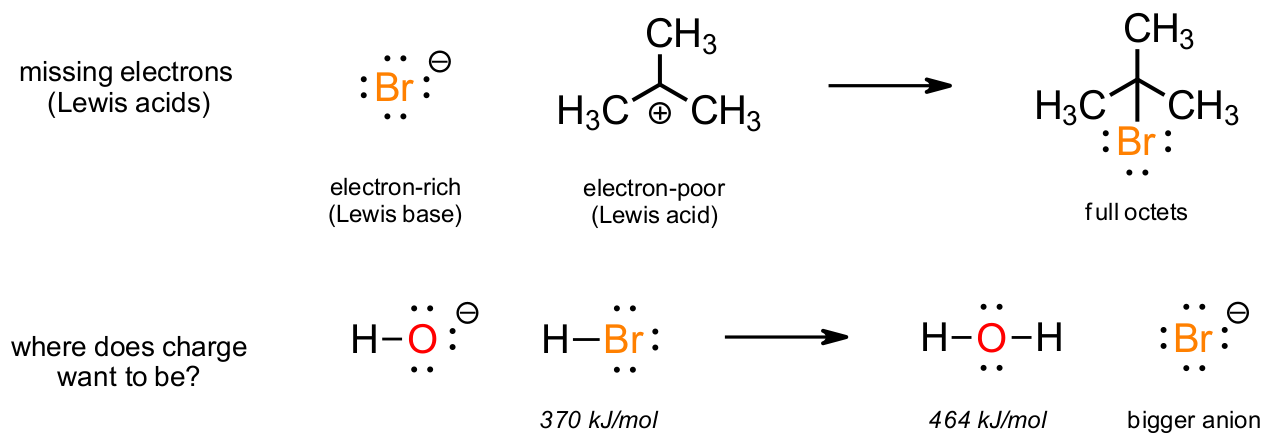
When an atom is missing electrons from the octet (e.g. B, Al, C+) they will be reactive towards electron-rich species. Positively charged carbon (i.e. carbocations) will feature regularly in Organic 1 and 2 and serve as electron-poor reactive intermediates. Their reactions as Lewis acids with electron-rich Lewis bases gives molecules with full octets. Also, when negative charge may be swapped from less stable (maybe smaller) atoms to one able to stabilize the charge better, this will result in predictable reactions.
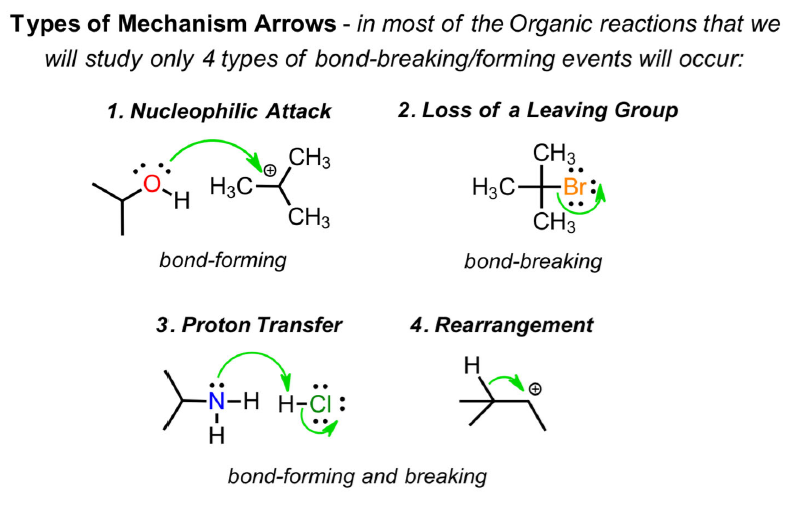
We use various devices for describing how chemical change takes place. Reaction profiles are helpful as they remind us of the energy changes that occur within, and motivation for, a chemical process. Mechanism arrows are an elegant way of describing how bonds are formed and broken along the way to products. The application of these ideas is discussed further here.